Assessing Physiological Response to Toxic Industrial Chemical Exposure in Megacities
Danielle L. Ippolito
Introduction
Megacities—urban areas with populations of ten million or greater—and other dense urban environments are emerging and growing globally, posing new challenges for U.S. military operations. The United States (U.S.) military can be better positioned for potential future operations within megacities as part of a joint, interagency, intergovernmental, and multinational team by increasing its understanding of megacity environments (Harris et al., 2014). Chemical exposures pose risk to the health and readiness of Service Members operating in these environments. The Naval Research Laboratory (NRL) prioritized a list of the top 30 chemicals of concern for global military operations based on physical characteristics and available toxicology data (Sutto, 2015). Occupational exposure to these chemicals increases risk of developing adverse health effects during mission operations or post-deployment. Policy guidelines specifying the use of the appropriate personal protective gear constitute the first line of defense to protect personnel by limiting exposure in the field. After a confirmed or suspected exposure event, far-forward diagnostic tools are needed to quickly and effectively determine and manage the risk of adverse health effects post-exposure in order to make informed command decisions about return-to-duty and treatment options. Identifying and quantifying biomolecular indicators in accessible biofluids such as blood, saliva, or urine is critically important for evaluating chemically-induced disease prognosis (Tawa et al., 2014; Ippolito et al., 2015). Biomarkers of end-organ toxicity can be integrated into a fieldable detection system to rapidly diagnose chemical exposure-induced adverse health effects in theater. Noninvasive or minimally invasive screening methods could enable early intervention, treatment, and informed decision making to optimize force readiness. Mapping biomolecular patterns of adverse health effects represents a promising solution to the complex problem of assessing health effects after exposure to mixtures of different chemicals and /or pollutants and aggregated exposure effects over time (Silins & Högberg, 2011).
This systematic literature review identifies and assesses the level of evidence for candidate far forward diagnostic technologies and biomarkers that may be used to detect emerging health effects from exposure to militarily-relevant chemicals. The review emphasizes a subacute exposure window (i.e., days to weeks) best suited to the short-term exposure scenarios anticipated in military operations and mission scenarios. Biomarker development demands meeting rigorous regulatory and clinical standards before adoption in the field, but military investment in this research and development process has the potential for significant returns in military healthcare during the deployment life cycle. Biomarker-based screening technologies can benefit routine health care screening, return-to-duty decision-making, triage during mass casualty exposure events, and/or guiding the development of diagnostics to inform treatment options. Biomarker-based far-forward diagnostic strategies and technologies have the potential to transform military healthcare in the changing face of warfare in megacities and dense urban operating environments
Methods
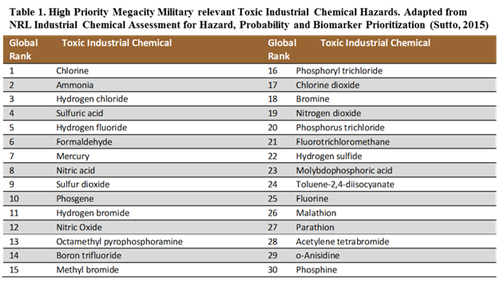
The peer-reviewed literature was surveyed to evaluate the weight-of-evidence regarding biomarkers for emerging health effects resulting from exposure to militarily-relevant chemicals (Table 1). The target list of militarily relevant chemicals was based on 30 prioritized megacity chemical hazards outlined in the NRL Industrial Chemical Assessment for Hazard, Probability, and Biomarker Prioritization (Sutto, 2015). A biomarker was defined as a molecular, cellular, or biophysical event linked to an emerging health effect.
Databases searched included PubMed, Web of Knowledge, Google Scholar, ClinicalTrials.gov, publicly available Department of Defense (DoD) technical reports (e.g., Defense Technical Information Center), non-DoD sources, and the Center for Disease Control Agency for Toxic Substance & Disease Registry and from the U.S. Library of Medicine Hazardous Substances Data Bank (“Agency for Toxic Substance & Disease Registry - Medical Management Guidelines Home Page,” n.d., “U.S. National Library of Medicine - Hazardous Substances Data Bank,” n.d.). Search terms were selected to identify studies that examined health effects associated with exposure to the high priority chemicals. The search was limited to articles published in English from January 2005 to May 2015.
Peer-reviewed articles were excluded if they (1) reported acute responses requiring immediate palliative care, (2) reviewed in vitro or computational toxicology sites that did not use publically available data sets, and (3) discussed mechanistic biomarkers without clear implications for candidate prognostic markers suitable for fieldable detection devices.
An objective two-step grading approach based on the U.S. Preventive Services Task Force Grade Definitions was used to assess the internal validity of individual studies published in peer-reviewed journal articles that met the inclusion criteria. The first step in evidence grading methodology identified and ranked the study design within a hierarchy of evidence, and the second step assessed the study quality (good, fair, poor). Study designs included the following categories: preclinical research (e.g., animal models), clinical research (e.g., clinical trials, diagnostic studies), epidemiological research (e.g., cohort studies, intervention studies), and secondary research (e.g., meta-analysis, systematic reviews). If a publication contained both a preclinical study and a clinical study, a separate grade was assigned for each study.
Results and Discussion
A review of 57 current papers in the diagnostic device literature identified five classes of biomarker detection devices (Figure 1). Most of the devices in development are lab-on-a-chip designs and paper-based lab-on-a-chip (LOC paper) protoypes. Frequently portable and disposable, these designs may be amenable to military field settings (Shafiee et al., 2015). Unique technological challenges limit the utility of many prototype diagnostics for use in a far-forward operational setting. Further research, clinical trials, and validation are needed to advance the devices beyond the prototype stage (Hoenigl et al., 2014). Ruggedization and miniaturization of laboratory prototypes are significant challenges in fielding these devices (Greenwood et al., 2007).
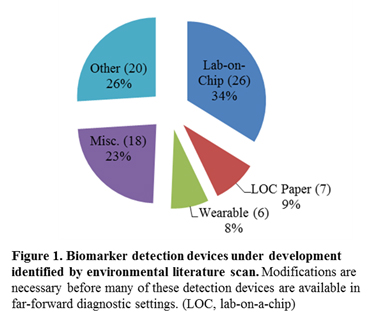
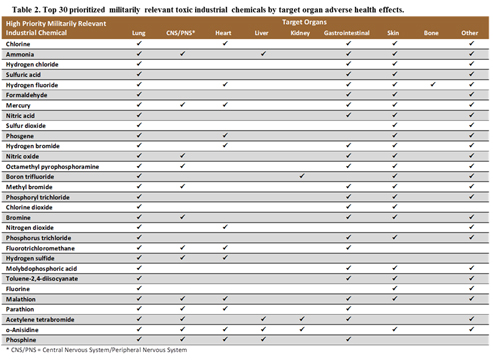
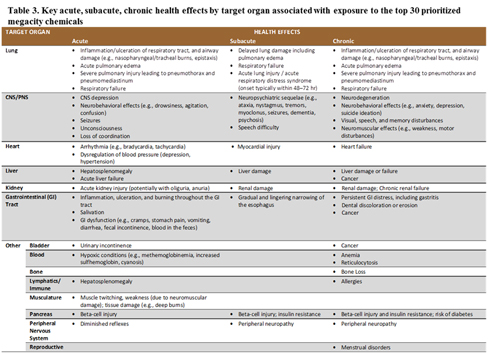
Table 2 lists the target organs of adverse health effects following exposure to each of the chemicals. Table 3 reports the key acute, subacute, and chronic health effects for each target organ. Most of the data summarized in Tables 2 and 3 are derived from a scan of public toxicology data repositories (e.g., the Hazardous Substances Data Bank and related ToxNet resources). These repositories identify relevant studies outside the time frame of the systematic review (i.e., before 2005-2015).
Articles meeting the inclusion criteria for adverse health effects were organized into eight specificntarget organ categories: lung, central nervous system (CNS), peripheral nervous system (PNS), heart, liver, kidney, gastrointestinal, other, and multiple (Figure 2). A summary of the literature review by target organ for five key target organs (lung, CNS/PNS, heart, liver, and kidney) follows.
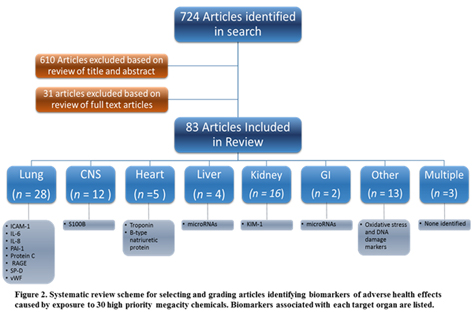
Lung. Several of the 28 lung-related articles identified biomarkers correlating with acute lung injury/acute respiratory distress syndrome (ALI/ARDS): RAGE, ICAM-1, KL-6, SP-D, vWF, IL-6, IL-8, protein C, PAI-1, TNFR1 and 2, and thrombomodulin (Agrawal et al., 2012; Calfee et al., 2008; Calfee et al., 2009; Collard et al., 2010; McClintock et al., 2008; Parsons et al., 2005; Uchida et al., 2006). Many of these biomarkers were associated with clinical outcomes (e.g., ventilator-free days, organ-failure-free days, and mortality). Mean levels the proteins PBEF or MIF were significantly greater in serum of ALI patients than healthy controls although the specificity for ALI remains uncertain (Gao et al., 2007; Ye et al., 2005). Combining predictive markers improved predictive power. Low levels of protein C and high levels of PAI-1 were independent predictors of mortality, and the two markers had a synergistic interaction for the risk of death (Ware et al., 2007). Levels of RAGE, PCP III, BNP, ANG-2, IL-8, TNF-α, and IL-10 show potential as a diagnostic biomarker panel (Fremont et al., 2010). In two studies, combinations of clinical predictors and multiple plasma biomarkers measured in individuals with ALI/ARDS improved predictive power for mortality over either approach alone, with some evidence that trauma differentially affects odds of mortality (Calfee et al., 2011; Ware et al., 2010). Exposure to isocyanates (e.g., TDI and MDI) was linked to occupational asthma. Biomarkers associated with this clinical outcome included VDBP, MMP-9, VEGF, LSP-1, COR1A, HPX, and autoantibodies to tTG, CK18, and CK19 (Kim et al., 2011; Hur et al., 2008; Ye et al., 2006; Haenen et al., 2012). F2-IsoP in plasma and lung tissue and SOD1 and COX2 in lung tissue may be associated with respiratory exposure to cadmium or silica nanoparticles (Coccini et al., 2012). Pulmonary fluid proteins and/or bronchial lavage proteins are biomarker candidates for lung injury, including ferritin, transferrin, CC16, ICAM-1, and PBEF (Hur et al, 2008; Kropski et al., 2009; Ye et al., 2005). Biomarkers in exhaled breath present an attractive alternative for accessibility in field settings (eNO and eCO2 after exposure to chlorine and phosgene) or cytokines such as IL-4 after exposure to NO2; Luo et al., 2014; Nath and Januszkiewicz, 2008).
Central and peripheral nervous system (CNS/PNS). The 18 articles associated with CNS/PNS injury included serum biomarkers associated with neurological sequelae, mostly associated with organophosphate pesticide (OP) exposure. These biomarkers of OP exposure and effect included a panel of autoantibodies (e.g., antibodies to NFP, TAU, MAP-2, MBP, GFAP, S100B), blood ChE/AChE, creatinine kinase, AST, LysoPC hydrolase in erythrocytes, SOD activity and/or LPO concentration after exposure to organophosphate pesticides (Abou-Donia et al., 2013; Rohlman et al., 2011; Bayrami et al., 2012; Vose et al., 2007; Colak et al., 2014; Aygun et al., 2007). Biomarkers to non-OP-related chemical exposures included GRIA 1 and glial S100B after exposure to mercury (Park et al., 2012) and serum S100B after acute CO poisoning (Abou-Donia, 2013).
Heart. A combination of biophysical and serum biomarkers were identified for the heart. Isoforms of troponin and cardiac natriuretic proteins released into the serum were associated with myocardial injury. Hydrogen sulfide poisoning resulted in cardiac proteins TNI and CPK-MB increased over time in conjunction with ECG data (Hirakawa et al., 2013). OP poisonings were associated with elevated TNI detecting acute early phase of cardiac injury (e.g., within the first 48 hours) (Cha, Cha et al., 2014). There is strong evidence for the emerging utility of cardiac troponins in stratifying toxicity risk (Dolci et al., 2008). Biophysical biomarkers included ECG measurements to determine risk of myocardial injury and arrhythmias (e.g., transmyocardial repolarization parameters to myocardial injury, left ventricle ejection fraction), especially after CO poisoning and OP exposure (Akilli et al., 2013). An emerging literature identifies micro-ribonucleic acids (miRNAs) in the prediction, detection, assessment, or treatment of myocardial damage and cardiac dysfunction (Sandhu & Maddock, 2014).
Liver. Four articles identified liver biomarkers in serum, including miRNAs (serum miR-125a-5p, miR-192 and miRNA-122; Zheng et al., 2015; Zhang et al., 2010). Changes in miRNA levels assessed in liver tissue implicate miRNAs such as miR-34a as promising new candidates in serum or urine (Koufaris et al., 2012). Liver injury diagnosis is routinely made in conjunction with clinical chemistry data, including alanine amino transferase (ALT). A panel of plasma or serum biomarkers diagnosed liver fibrosis in animal models (fibrinogen precursor, ceruloplasmin isoform 1, insulin like growth factor binding protein, alpha-2-macroglobulin, and vitronectin) (Ippolito et al., 2015).
Kidney. The 17 kidney-related articles identified biomarkers in serum and urine. Acute kidney injury markers in serum included LG3, cathepsin L, NGAL, IL-6, soluble TNFR1 and 2, and PAI-1 as potential candidates (Haase et al, 2014; Liu et al., 2007). Clinical studies have identified promising urinary biomarkers of acute kidney injury and/or nephrotoxicity, including KIM-1 protein after TCE exposure (Vermeulen et al., 2012). Urinary panels include Alb, NAG and α1-MG as indicators of fluoride and arsenic-induced glomerular and tubular injury (Zeng et al., 2014). KIM-1 predicted kidney injury earlier than the traditional biomarkers, such as creatinine, BUN, and/or NGAL (Rached et al., 2008; Vaidya et al., 2010; Wunnapuk, Gobe, et al., 2014; Zhou et al., 2008). Many of the preclinical studies examined urinary biomarkers of kidney injury other than KIM-1 (e.g., Cal, Clu, KIM‑1, Lcn2, the three-branched chain amino acids [leucine, isoleucine, and valine], OPN, TTF3) and provide important foundational information (Boudonck et al., 2009; Fuchs et al., 2014; Hoffmann et al., 2010; Wunnapuk, Liu, et al., 2014; Yu et al., 2010).
Other health effects. Other health effects with biomarker candidates included gastrointestinal, reproductive, cancer progression, blood, bone, and lymphatics (see Tables 2 and 3 and Figure 2).
Conclusions
As megacities and dense urban environments continue to grow in number and population size, there is an unmet need to understand the health threats of service members exposed to toxic chemicals and environmental hazards unique to the megacity operational environment. Properly validated biomarkers of exposure and effect can integrate with current biomonitoring systems throughout the soldier deployment life cycle to inform medical decisions during and after exposure events, especially in polluted megacity operational environments with increased risk of non-agent chemical exposure. Further research is needed to ensure the fieldability of ruggedized detection systems in the megacity environment.
Acknowledgements: This manuscript is based on a DTIC report submitted by Booz Allen Hamilton entitled “Biomarkers for Health Effects of Industrial Chemical Exposure Literature Review”, submitted to USACEHR on behalf of USAMRMC-MOMRP on 26 June 2015. We gratefully acknowledge COL Thomas C. Timmes, USA, Commander of USACEHR, for programmatic support.
The views, opinions, and/or findings contained in this report are those of the author and should not be construed as official Department of the Army position, policy, or decision, unless so designated by other official documentation.
Acronyms and Abbreviations
AChE Acetylcholinesterase
Alb Albumin
ALI Acute Lung Injury
ALP Alkaline Phosphatase
ALT Alanine Aminotransferase
ANG-2 Angiopoitetin-2
ARDS Acute Respiratory Distress Syndrome
AST Aspartate Aminotransferase
BNP B-type Natriuretic Protein
BUN Blood Urea Concentration
Cal Calbindin-D28
CC16 Clara Cell 16
ChAT Choline Acetyltransferase
ChE Cholinesterase
CI Confidence Interval
CK Cytokeratin
Clu Clusterin
CNS Central Nervous System
CO Carbon Monoxide
COR1A Coronin 1A
COX2 Cyclooxygenase Type 2
CPK Creatine Kinase
DNA Deoxyribonucleic acid
ECG Electrocardiogram
eCO2 Exhaled Carbon Dioxide
eNO Exhaled Nitric Oxide
F2-IsoP F2-Isoprostanes
GFAP Glial Fibrillary Acidic Protein
GRIA 1 Glutamate Receptor 1
HPX Hemopexin
HSA Human Serum Albumin
ICAM-1 Intercellular Adhesion Molecule-1
IL Interleukin
KIM-1 Kidney Injury Molecule-1
Lcn2 Lipocalin 2
LG3 Prelecan C-terminal Fragment LG3
LPO Lipid Peroxidation
LSP-1 Lymphocyte Specific Protein-1
LysoPC Lysophosphatidylcholine
MBP Myelin Basic Protein
MDI Methylene Diphenyl Diisocyanate
MIF Migration Inhibitory Factor
miRNA micro-Ribonucleic Acid
MMP-9 Matrix Metalloproteinase-9
MPO Myeloperoxidase
NAG N-Acetyl-β-D-Glucosaminidase
NFP Neurofilament Triplet Proteins
NGAL Neutrophil Gelatinase-associated Lipocalin
NMP22 Nuclear Matrix Protein-22
NO2 Nitrogen Dioxide
OP Organophosphate
PAI-1 Plasminogen Activator Inhibitor-1
PBEF Pre-B-Cell Colony-Enhancing Factor
PCP III Procollagen Peptide III
PNS Peripheral Nervous System
RAGE Receptor for Advanced Glycation Endproducts
S100B S100 Calcium-Binding Protein B
SOD Superoxide Dismutase
SP Surfactant Protein
TCE Trichloroethylene
TDI Toulene-2,4-Diisocynate
TFF3 Trefoil Factor 3
TIC Toxic industrial chemical
TIM Toxic industrial material
TNFR Tumor Necrosis Factor Receptor
TNI Troponin I
Tp-e Tpeak-Tend
U.S. United States
USACEHR United States Army Center for Environmental Health Research
VDBP Vitamin-D-Binding Protein
VEGF Vascular Endothelial Growth Factor
vWF von Willebrand Factor
α1-MG α-1-microglobulin
References
- Abou-Donia, M. B., Abou-Donia, M. M., ElMasry, E. M., Monro, J. A., & Mulder, M. F. A. (2013). Autoantibodies to Nervous System-Specific Proteins Are Elevated in Sera of Flight Crew Members: Biomarkers for Nervous System Injury. Journal of Toxicology and Environmental Health, Part A, 76(6), 363–380. http://doi.org/10.1080/15287394.2013.765369
- Agrawal, A., Zhuo, H., Brady, S., Levitt, J., Steingrub, J., Siegel, M. D., … Liu, K. D. (2012). Pathogenetic and predictive value of biomarkers in patients with ALI and lower severity of illness: results from two clinical trials. American Journal of Physiology - Lung Cellular and Molecular Physiology, 303(8), L634–L639. http://doi.org/10.1152/ajplung.00195.2012
- Akilli, N. B., Akinci, E., Akilli, H., Dundar, Z. D., Koylu, R., Polat, M., & Cander, B. (2013). A new marker for myocardial injury in carbon monoxide poisoning: T peak-T end. The American Journal of Emergency Medicine, 31(12), 1651–1655. http://doi.org/10.1016/j.ajem.2013.08.049
- Aygun, D., Erenler, A. K., Karatas, A. D., & Baydin, A. (2007). Intermediate Syndrome Following Acute Organophosphate Poisoning: Correlation with Initial Serum Levels of Muscle Enzymes*. Basic & Clinical Pharmacology & Toxicology, 100(3), 201–204. http://doi.org/10.1111/j.1742-7843.2007.00042.x
- Bayrami, M., Hashemi, T., Malekirad, A. A., Ashayeri, H., Faraji, F., & Abdollahi, M. (2012). Electroencephalogram, cognitive state, psychological disorders, clinical symptom, and oxidative stress in horticulture farmers exposed to organophosphate pesticides. Toxicology and Industrial Health, 28(1), 90–96. http://doi.org/10.1177/0748233711407243
- Boudonck, K. J., Rose, D. J., Karoly, E. D., Lee, D. P., Lawton, K. A., & Lapinskas, P. J. (2009). Metabolomics for early detection of drug-induced kidney injury: review of the current status. Bioanalysis, 1(9), 1645–1663. http://doi.org/10.4155/bio.09.142
- Calfee, C. S., Eisner, M. D., Ware, L. B., Thompson, B. T., Parsons, P. E., Wheeler, A. P., … Matthay, M. A. (2007). Trauma-associated lung injury differs clinically and biologically from acute lung injury due to other clinical disorders. Critical Care Medicine, 35(10), 2243–2250.
- Calfee, C. S., Ware, L. B., Eisner, M. D., Parsons, P. E., Thompson, B. T., Wickersham, N., … NHLBI ARDS Network. (2008). Plasma receptor for advanced glycation end products and clinical outcomes in acute lung injury. Thorax, 63(12), 1083–1089. http://doi.org/10.1136/thx.2008.095588
- Calfee, C. S., Ware, L. B., Glidden, D. V., Eisner, M. D., Parsons, P. E., Thompson, B. T., & Matthay, M. A. (2011). Use of risk reclassification with multiple biomarkers improves mortality prediction in acute lung injury. Critical Care Medicine, 39(4), 711–717. http://doi.org/10.1097/CCM.0b013e318207ec3c
- Cha, Y. S., Cha, K. C., Kim, O. H., Lee, K. H., Hwang, S. O., & Kim, H. (2014). Features and predictors of myocardial injury in carbon monoxide poisoned patients. Emergency Medicine Journal: EMJ, 31(3), 210–215. http://doi.org/10.1136/emermed-2012-202152
- Cha, Y. S., Kim, H., Go, J., Kim, T. H., Kim, O. H., Cha, K. C., … Hwang, S. O. (2014). Features of myocardial injury in severe organophosphate poisoning. Clinical Toxicology (Philadelphia, Pa.), 52(8), 873–879. http://doi.org/10.3109/15563650.2014.944976
- Coccini, T., Roda, E., Manzo, L., Barni, S., & Signorini, C. (2012). Isoprostanes as Biomarkers for In Vivo Evaluation of Nanoparticle-induced Oxidative Stress: a Study with Silica Nanoparticles Doped with Cadmium. International Journal of Theoretical and Applied Nanotechnology. http://doi.org/10.11159/ijtan.2012.001
- Çolak, Ş., Erdogan, M. Ö., Baydin, A., Afacan, M. A., Kati, C., & Duran, L. (2014). Epidemiology of organophosphate intoxication and predictors of intermediate syndrome. Retrieved from http://dergipark.ulakbim.gov.tr/tbtkmedical/article/viewFile/5000022935/5000023173
- Collard, H. R., Calfee, C. S., Wolters, P. J., Song, J. W., Hong, S.-B., Brady, S., … others. (2010). Plasma biomarker profiles in acute exacerbation of idiopathic pulmonary fibrosis. American Journal of Physiology-Lung Cellular and Molecular Physiology, 299(1), L3–L7.
- Dolci, A., Dominici, R., Cardinale, D., Sandri, M. T., & Panteghini, M. (2008). Biochemical markers for prediction of chemotherapy-induced cardiotoxicity: systematic review of the literature and recommendations for use. American Journal of Clinical Pathology, 130(5), 688–695. http://doi.org/10.1309/AJCPB66LRIIVMQDR
- Fuchs, T. C., Mally, A., Wool, A., Beiman, M., & Hewitt, P. (2014). An Exploratory Evaluation of the Utility of Transcriptional and Urinary Kidney Injury Biomarkers for the Prediction of Aristolochic Acid–Induced Renal Injury in Male Rats. Veterinary Pathology Online, 51(3), 680–694. http://doi.org/10.1177/0300985813498779
- Gao, L., Flores, C., Fan-Ma, S., Miller, E. J., Moitra, J., Moreno, L., … Garcia, J. G. N. (2007). Macrophage migration inhibitory factor in acute lung injury: expression, biomarker, and associations. Translational Research, 150(1), 18–29. http://doi.org/10.1016/j.trsl.2007.02.007
- Greenwood, D. Q. (2007). Reconstructing operational theory: A framework for emerging threats in a complex environment. Marine Corps University, School of Advanced Warfighting. Quantico, VA.
- Haase, M., Bellomo, R., Albert, C., Vanpoucke, G., Thomas, G., Laroy, W., … Haase-Fielitz, A. (2014). The identification of three novel biomarkers of major adverse kidney events. Biomarkers in Medicine, 8(10), 1207–1217. http://doi.org/10.2217/bmm.14.90
- Haenen, S., Clynen, E., De Vooght, V., Schoofs, L., Nemery, B., Hoet, P. H. M., & Vanoirbeek, J. A. J. (2012). Proteome changes in auricular lymph nodes and serum after dermal sensitization to toluene diisocyanate in mice. Proteomics, 12(23-24), 3548–3558. http://doi.org/10.1002/pmic.201200264
- Harris, M., Dixon, R., Melin, N., Hendrex, D., Russo, R., & Bailey, M. (2014). Megacities and the United States Army: Preparing for a Complex and Uncertain Future.
- Hirakawa, A., Takeyama, N., Iwatsuki, S., Iwata, T., & Kano, H. (2013). Delayed myocardial injury following acute hydrogen sulfide intoxication. Chūdoku Kenkyū: Chūdoku Kenkyūkai Jun Kikanshi = The Japanese Journal of Toxicology, 26(1), 44–48.
- Hoenigl, M., Prattes, J., Spiess, B., Wagner, J., Prueller, F., Raggam, R. B., … Buchheidt, D. (2014). Performance of galactomannan, Beta-d-Glucan, aspergillus lateral-flow device, conventional culture, and PCR tests with bronchoalveolar lavage fluid for diagnosis of invasive pulmonary aspergillosis. Journal of Clinical Microbiology, 52(6), 2039–2045. http://doi.org/10.1128/JCM.00467-14
- Hoffmann, D., Adler, M., Vaidya, V., Rached, E., Mulrane, L., Gallagher, W. M., … Mally, A. (2010). Performance of novel kidney biomarkers in preclinical toxicity studies. Toxicological Sciences, kfq029. http://doi.org/10.1093/toxsci/kfq029
- Hur, G.-Y., Choi, G.-S., Sheen, S.-S., Lee, H.-Y., Park, H.-J., Choi, S.-J., … Park, H.-S. (2008). Serum ferritin and transferrin levels as serologic markers of methylene diphenyl diisocyanate–induced occupational asthma. Journal of Allergy and Clinical Immunology, 122(4), 774–780.
- Ippolito DL, AbdulHameed MDM, Tawa GJ, Baer CE, Boyle MH, Hobbs CA, Streicker MA, Snowden BS, Lewis JA, Wallqvist A, Stallings JD. Gene expression patterns associated with histopathology in toxic liver injury. Toxicological Sciences. 2016 149(1):67-88. doi: 10.1093/toxsci/kfv214.
- Kim, J.-H., Kim, J.-E., Choi, G.-S., Kim, H.-Y., Ye, Y.-M., & Park, H.-S. (2011). Serum cytokines markers in toluene diisocyanate-induced asthma. Respiratory Medicine, 105(7), 1091–1094. http://doi.org/10.1016/j.rmed.2011.03.005
- Koufaris, C., Wright, J., Currie, R. A., & Gooderham, N. J. (2012). Hepatic microRNA profiles offer predictive and mechanistic insights after exposure to genotoxic and epigenetic hepatocarcinogens. Toxicological Sciences, kfs170. http://doi.org/10.1093/toxsci/kfs170
- Kropski, J. A., Fremont, R. D., Calfee, C. S., & Ware, L. B. (2009). Clara cell protein (cc16), a marker of lung epithelial injury, is decreased in plasma and pulmonary edema fluid from patients with acute lung injury. Chest, 135(6), 1440–1447. http://doi.org/10.1378/chest.08-2465
- Liu, K. D., Glidden, D. V., Eisner, M. D., Parsons, P. E., Ware, L. B., Wheeler, A., … Matthay, M. A. (2007). Predictive and pathogenetic value of plasma biomarkers for acute kidney injury in patients with acute lung injury. Critical Care Medicine, 35(12), 2755–2761.
- Luo, S., Trübel, H., Wang, C., & Pauluhn, J. (2014). Phosgene- and chlorine-induced acute lung injury in rats: comparison of cardiopulmonary function and biomarkers in exhaled breath. Toxicology, 326, 109–118. http://doi.org/10.1016/j.tox.2014.10.010
- McClintock, D., Zhuo, H., Wickersham, N., Matthay, M. A., & Ware, L. B. (2008). Biomarkers of inflammation, coagulation and fibrinolysis predict mortality in acute lung injury. Critical Care, 12(2), R41. http://doi.org/10.1186/cc6846
- Nath, J., & Januszkiewicz, A. (2008). Early Systemic Biomarkers of Acute Lung Injury: Application of Multiplex Proteomic Array Technology. Walter Reed Army Institute of Research Silver Spring MD. Retrieved from http://www.dtic.mil/docs/citations/ADA505713
- Park, E., Ahn, J., Min, Y.-G., Jung, Y.-S., Kim, K., Lee, J., & Choi, S.-C. (2012). The usefulness of the serum s100b protein for predicting delayed neurological sequelae in acute carbon monoxide poisoning. Clinical Toxicology, 50(3), 183–188. http://doi.org/10.3109/15563650.2012.658918
- Parsons, P. E., Matthay, M. A., Ware, L. B., Eisner, M. D., & National Heart, Lung, Blood Institute Acute Respiratory Distress Syndrome Clinical Trials Network. (2005). Elevated plasma levels of soluble TNF receptors are associated with morbidity and mortality in patients with acute lung injury. American Journal of Physiology. Lung Cellular and Molecular Physiology, 288(3), L426–431. http://doi.org/10.1152/ajplung.00302.2004
- Rached, E., Hoffmann, D., Blumbach, K., Weber, K., Dekant, W., & Mally, A. (2008). Evaluation of Putative Biomarkers of Nephrotoxicity after Exposure to Ochratoxin A In Vivo and In Vitro. Toxicological Sciences, 103(2), 371–381. http://doi.org/10.1093/toxsci/kfn040
- Rohlman, D. S., Anger, W. K., & Lein, P. J. (2011). Correlating neurobehavioral performance with biomarkers of organophosphorous pesticide exposure. Neurotoxicology, 32(2), 268–276. http://doi.org/10.1016/j.neuro.2010.12.008
- Sandhu, H., & Maddock, H. (2014). Molecular basis of cancer-therapy-induced cardiotoxicity: introducing microRNA biomarkers for early assessment of subclinical myocardial injury. Clinical Science (London, England: 1979), 126(6), 377–400. http://doi.org/10.1042/CS20120620
- Shafiee, H., Asghar, W., Inci, F., Yuksekkaya, M., Jahangir, M., Zhang, M. H., … Demirci, U. (2015). Paper and flexible substrates as materials for biosensing platforms to detect multiple biotargets. Scientific Reports, 5. http://doi.org/10.1038/srep08719
- Silins, I., & Högberg, J. (2011). Combined Toxic Exposures and Human Health: Biomarkers of Exposure and Effect. International Journal of Environmental Research and Public Health, 8(3), 629–647. http://doi.org/10.3390/ijerph8030629
- Singh, S., Kumar, V., Thakur, S., Banerjee, B. D., Chandna, S., Rautela, R. S., … Rai, A. (2011). DNA damage and cholinesterase activity in occupational workers exposed to pesticides. Environmental Toxicology and Pharmacology, 31(2), 278–285. http://doi.org/10.1016/j.etap.2010.11.005
- Sutto, T. (2015). NRL Industrial Chemical Assessment for Hazard, Probability and Biomarker Prioritization (No. NRL FR 6364-14-14, XXX) (pp. 1–35). Washington, DC: Naval Research Laboratory.
- Tawa, G. J., AbdulHameed, M. D. M., Yu, X., Kumar, K., Ippolito, D. L., Lewis, J. A., … Wallqvist, A. (2014). Characterization of Chemically Induced Liver Injuries Using Gene Co-Expression Modules. PLoS ONE, 9(9), e107230. http://doi.org/10.1371/journal.pone.0107230
- Uchida, T., Shirasawa, M., Ware, L. B., Kojima, K., Hata, Y., Makita, K., … Matthay, M. A. (2006). Receptor for Advanced Glycation End-Products Is a Marker of Type I Cell Injury in Acute Lung Injury. American Journal of Respiratory and Critical Care Medicine, 173(9), 1008–1015. http://doi.org/10.1164/rccm.200509-1477OC
- U.S. National Library of Medicine - Hazardous Substances Data Bank. (n.d.). Retrieved June 9, 2015, from http://toxnet.nlm.nih.gov/cgi-bin/sis/htmlgen?HSDB
- Vaidya, V. S., Ozer, J. S., Dieterle, F., Collings, F. B., Ramirez, V., Troth, S., … Bonventre, J. V. (2010). Kidney injury molecule-1 outperforms traditional biomarkers of kidney injury in preclinical biomarker qualification studies. Nature Biotechnology, 28(5), 478–485. http://doi.org/10.1038/nbt.1623
- Vermeulen, R., Zhang, L., Spierenburg, A., Tang, X., Bonventre, J. V., Reiss, B., … Lan, Q. (2012). Kidney toxicity in Chinese factory workers exposed to Trichloroethylene. Carcinogenesis, bgs191. http://doi.org/10.1093/carcin/bgs191
- Vose, S. C., Holland, N. T., Eskenazi, B., & Casida, J. E. (2007). Lysophosphatidylcholine hydrolases of human erythrocytes, lymphocytes, and brain: Sensitive targets of conserved specificity for organophosphorus delayed neurotoxicants. Toxicology and Applied Pharmacology, 224(1), 98–104. http://doi.org/10.1016/j.taap.2007.06.008
- Ware, L. B., Koyama, T., Billheimer, D. D., Wu, W., Bernard, G. R., Thompson, B. T., … Matthay, M. A. (2010). Prognostic and pathogenetic value of combining clinical and biochemical indices in patients with acute lung injury. Chest, 137(2), 288–296. http://doi.org/10.1378/chest.09-1484
- Ware, L. B., Matthay, M. A., Parsons, P. E., Thompson, B. T., Januzzi, J. L., Eisner, M. D., & National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome Clinical Trials Network. (2007). Pathogenetic and prognostic significance of altered coagulation and fibrinolysis in acute lung injury/acute respiratory distress syndrome. Critical Care Medicine, 35(8), 1821–1828. http://doi.org/10.1097/01.CCM.0000221922.08878.49
- Wunnapuk, K., Gobe, G., Endre, Z., Peake, P., Grice, J. E., Roberts, M. S., … Liu, X. (2014). Use of a glyphosate-based herbicide-induced nephrotoxicity model to investigate a panel of kidney injury biomarkers. Toxicology Letters, 225, 192–200.
- Wunnapuk, K., Liu, X., Gobe, G. C., Endre, Z. H., Peake, P. W., Grice, J. E., … Buckley, N. A. (2014). Kidney biomarkers in MCPA-induced acute kidney injury in rats: Reduced clearance enhances early biomarker performance. Toxicology Letters, 225, 467–478.
- Ye, S. Q., Simon, B. A., Maloney, J. P., Zambelli-Weiner, A., Gao, L., Grant, A., … Garcia, J. G. N. (2005). Pre–B-Cell Colony-enhancing Factor as a Potential Novel Biomarker in Acute Lung Injury. American Journal of Respiratory and Critical Care Medicine, 171(4), 361–370. http://doi.org/10.1164/rccm.200404-563OC
- Ye, Y.-M., Nahm, D.-H., Kim, C.-W., Kim, H.-R., Hong, C.-S., Park, C.-S., … Park, H.-S. (2006). Cytokeratin autoantibodies: useful serologic markers for toluene diisocyanate-induced asthma. Yonsei Medical Journal, 47(6), 773–781.
- Yu, Y., Jin, H., Holder, D., Ozer, J. S., Villarreal, S., Shughrue, P., … Gerhold, D. L. (2010). Urinary biomarkers trefoil factor 3 and albumin enable early detection of kidney tubular injury. Nature Biotechnology, 28(5), 470–477. http://doi.org/10.1038/nbt.1624
- Zeng, Q., Xu, Y., Yu, X., Yang, J., Hong, F., & Zhang, A. (2014). The combined effects of fluorine and arsenic on renal function in a Chinese population. Toxicology Research, 3(5), 359–366. http://doi.org/10.1039/C4TX00038B
- Zhang, Y., Liu, X., McHale, C., Li, R., Zhang, L., Wu, Y., … Ding, S. (2013). Bone marrow injury induced via oxidative stress in mice by inhalation exposure to formaldehyde. PloS One, 8(9), e74974. http://doi.org/10.1371/journal.pone.0074974
- Zheng, J., Zhou, Z., Xu, Z., Li, G., Dong, P., Chen, Z., … Yu, F. (2015). Serum microRNA-125a-5p, a useful biomarker in liver diseases, correlates with disease progression. Molecular Medicine Reports. http://doi.org/10.3892/mmr.2015.3546
- Zhou, Y., Vaidya, V. S., Brown, R. P., Zhang, J., Rosenzweig, B. A., Thompson, K. L., … Goering, P. L. (2008). Comparison of Kidney Injury Molecule-1 and Other Nephrotoxicity Biomarkers in Urine and Kidney Following Acute Exposure to Gentamicin, Mercury, and Chromium. Toxicological Sciences, 101(1), 159–170. http://doi.org/10.1093/toxsci/kfm260
